Description
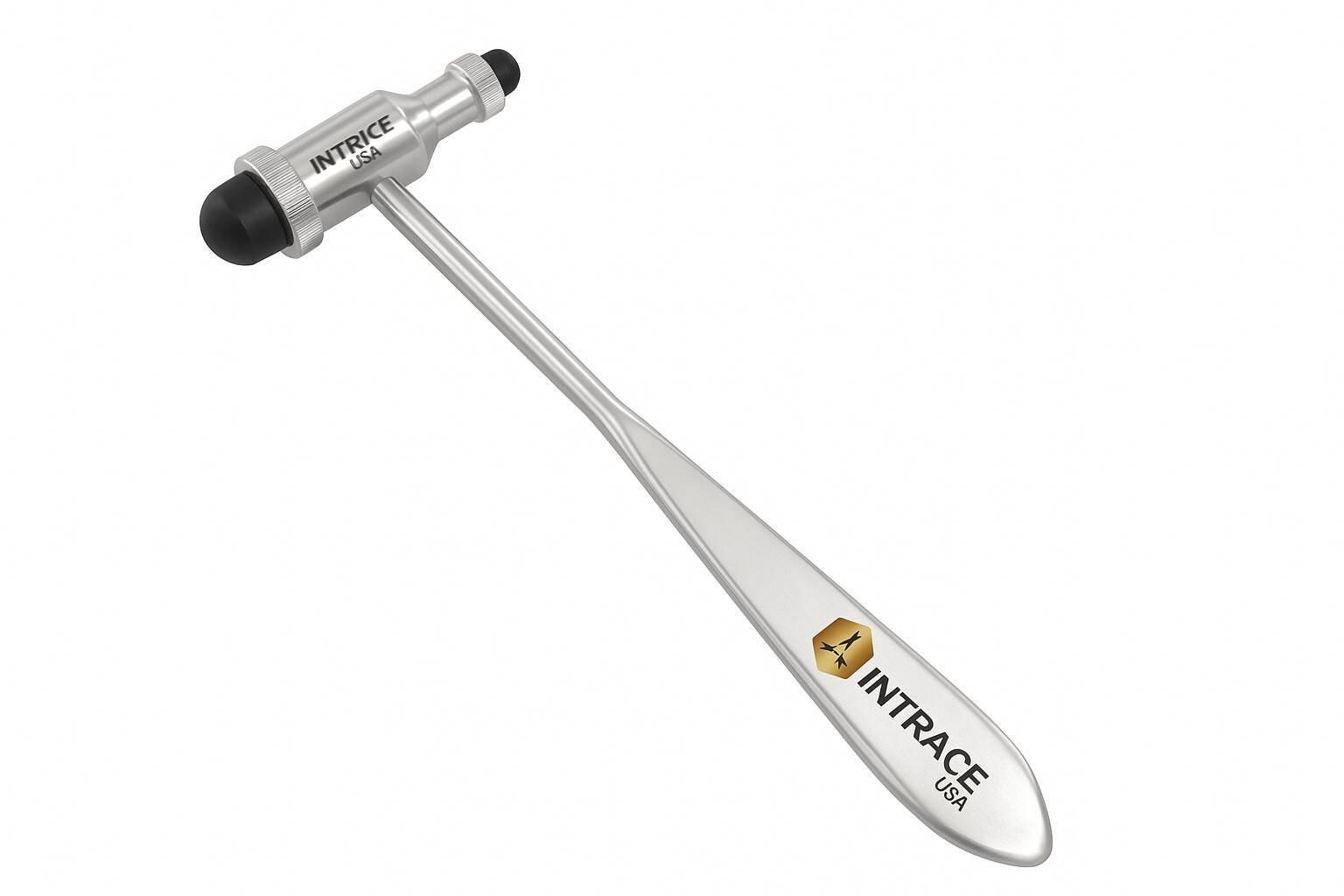
The Koenig Tuning Fork by Intrace USA provides otolaryngologists and audiologists with a reliable instrument for conducting essential hearing and sensory assessments. Because this Koenig Diagnostic Tuning Fork is precision-calibrated to specific frequencies, clinicians achieve accurate results when performing Rinne and Weber tests to differentiate between conductive and sensorineural hearing loss. Furthermore, Intrace USA manufactures every unit from premium surgical-grade stainless steel to ensure lasting mechanical integrity and consistent acoustic resonance. As a result, each unit undergoes a rigorous multi-point inspection at our U.S. facility to guarantee professional-grade reliability in every diagnostic environment.
Technical Features of the Koenig Diagnostic Tuning Fork:
-
The fixed-frequency prongs are specifically engineered to deliver a pure, sustained tone for consistent auditory and vibratory testing.
-
A balanced, ergonomic handle allows the examiner to maintain a secure grip while applying the base to bony prominences.
-
In addition, high-quality surgical-grade stainless steel construction ensures the instrument remains resistant to corrosion and frequency drift over time.
-
This Koenig Diagnostic Tuning Fork features a smooth, polished finish that is easy to sanitize between clinical examinations.
Common Applications in Auditory Testing:
ENT specialists and general practitioners primarily use this instrument to evaluate air and bone conduction thresholds in patients with suspected hearing impairments. Moreover, the standardized weight and geometry provide the examiner with predictable oscillation patterns for reliable data collection. Therefore, it serves as a critical tool for medical professionals who prioritize diagnostic accuracy, patient care, and procedural efficiency.
Maintenance for the Koenig Diagnostic Tuning Fork:
To maintain long-term acoustic performance, clean the instrument with a neutral pH detergent and a soft cloth to remove skin oils and debris. Consequently, you should store the tool in a protective case when not in use to prevent nicks or structural damage that could alter the frequency. However, avoid striking the prongs against hard metal surfaces, as this can compromise the calibrated resonance of the instrument.
Compliance and Quality:
Intrace USA manufactures this product in accordance with FDA, CE, and ISO 13485 quality standards. Therefore, we inspect each unit before distribution to ensure it meets professional diagnostic requirements. For more information on our quality standards, please visit our FAQs. To view our full range of auditory tools, please visit our Diagnostic Medical Instruments category page.
Disclaimer:
We provide images for reference only. Consequently, these may include multiple frequency variations (e.g., 256 Hz, 512 Hz). Please review all details carefully before ordering. Professionals should use these products. Because they contain stainless steel, they also contain nickel. Therefore, Intrace USA assumes no liability for allergic reactions or adverse effects related to material sensitivities. For additional safety data, refer to the FDA guidelines on medical device materials.








Reviews
There are no reviews yet.