Description
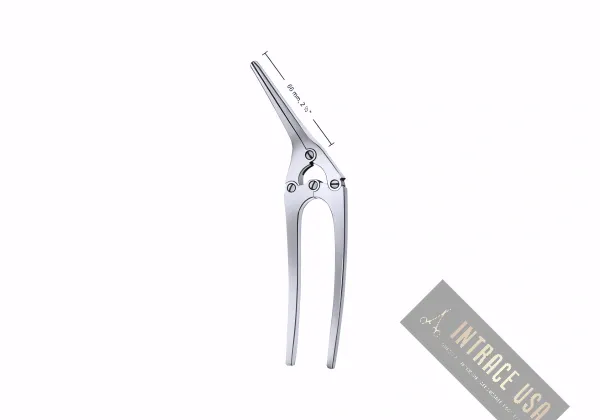
The Payr Intestinal and Stomach Clamp by Intrace USA provides general surgeons with a heavy-duty, high-precision instrument for secure gastric and intestinal occlusion. Because this Payr Intestinal Clamp features a robust crushing mechanism and an integrated alignment pin, clinicians achieve a definitive, leak-proof seal on thick tissue segments. Furthermore, Intrace USA manufactures every unit from premium surgical-grade stainless steel to ensure lasting mechanical integrity and maximum resistance to high-torque maneuvers. As a result, each unit undergoes a rigorous multi-point inspection at our U.S. facility to guarantee professional-grade reliability and precision in every operating room.
Technical Features of the Payr Intestinal Clamp:
-
The integrated alignment pin ensures the jaws remain perfectly parallel, providing uniform crushing pressure across the entire width of the stomach or bowel.
-
Longitudinal jaw serrations provide superior traction to prevent tissue “milking” or slippage during high-pressure occlusion.
-
In addition, high-quality surgical-grade stainless steel construction ensures maximum corrosion resistance and durability.
-
This Payr Intestinal Clamp remains fully autoclavable and complies with standard clinical sterilization protocols.
Primary Clinical Applications for this Surgical Tool:
General and GI surgeons primarily use the Payr Intestinal Clamp for the total occlusion of the stomach and intestinal walls during resection and reconstruction. Moreover, the high-leverage handle design allows the surgeon to apply controlled, heavy-duty pressure, ensuring a bloodless and sterile surgical field. Therefore, it serves as a critical tool for medical professionals who prioritize mechanical security, procedural efficiency, and instrument longevity.
Maintenance Guidelines for Your Intestinal Instruments:
To maintain long-term performance, clean the instrument with a neutral pH detergent immediately after use, paying close attention to the alignment pin and the deep longitudinal serrations. Consequently, you should follow standard facility autoclaving procedures to ensure the tool remains sterile. However, avoid abrasive pads or harsh chemicals that could compromise the specialized surgical finish.
Compliance and Quality:
Intrace USA manufactures this product in accordance with FDA, CE, and ISO 13485 quality standards. Therefore, we inspect each unit before distribution to ensure it meets professional clinical requirements. For more information on our quality standards, please visit our FAQs. To view our full range of specialized instruments, please visit our Intestinal and Rectal category page.
Disclaimer:
We provide images for reference only. Consequently, these may include multiple variations or lengths. Please review all details carefully before ordering. Professionals should use these products. Because they contain stainless steel, they also contain nickel. Therefore, Intrace USA assumes no liability for allergic reactions or adverse effects related to material sensitivities. For additional safety data, refer to the FDA guidelines on medical device materials.








Reviews
There are no reviews yet.