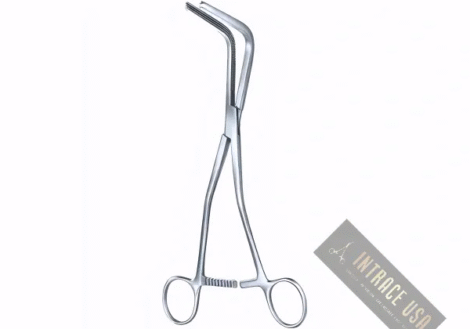
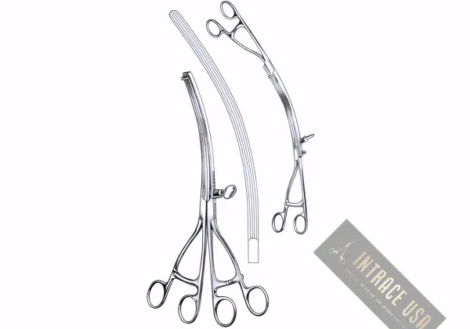
Description
The Ives-Fansler Anoscope by Intrace USA provides colorectal specialists with a specialized diagnostic and operative tool for the clear visualization of the anal canal. Because this instrument features a precision-fitted obturator and a high-polish interior barrel, clinicians achieve exceptionally smooth, atraumatic insertion while maintaining superior light reflection. Furthermore, Intrace USA manufactures every Ives-Fansler Anoscope from premium surgical-grade stainless steel to ensure lasting mechanical integrity and structural strength. As a result, each unit undergoes a rigorous multi-point inspection at our U.S. facility to guarantee professional-grade clarity and performance in every clinical setting.
Key Features of the Ives-Fansler Anoscope:
-
The smooth, bullet-tipped obturator ensures patient comfort during initial entry and placement.
-
A hollow, tubular design provides an unobstructed path for diagnostic swabs, biopsies, or local treatments.
-
In addition, high-quality surgical-grade stainless steel construction ensures long-term durability and maximum corrosion resistance.
-
This Ives-Fansler Anoscope remains fully autoclavable and complies with standard clinical sterilization protocols.
Clinical Applications for the Ives-Fansler Anoscope:
Gastroenterologists and surgeons primarily use the Ives-Fansler Anoscope for routine diagnostic inspections, hemorrhoid ligation, and internal tissue evaluations. Moreover, the ergonomic handle design provides the examiner with a stable, non-slip grip for controlled manipulation and rotation during the procedure. Therefore, it serves as a critical tool for medical professionals who prioritize procedural accuracy, patient safety, and reliable diagnostic outcomes.
Care and Maintenance:
To maintain long-term performance, clean the instrument with a neutral pH detergent immediately after use, ensuring all biological residue is removed from the barrel and obturator. Consequently, you should follow standard facility autoclaving procedures to ensure the tool remains sterile. However, avoid abrasive pads or harsh chemicals that could compromise the specialized surgical finish.
Compliance and Quality:
Intrace USA manufactures this product in accordance with FDA, CE, and ISO 13485 quality standards. Therefore, we inspect each unit before distribution to ensure it meets professional clinical requirements. For more information on our quality standards, please visit our FAQs. To view our full range of specialized instruments, please visit our Intestinal and Rectal category page.
Disclaimer:
We provide images for reference only. Consequently, these may include multiple sizes or variations. Please review all details carefully before ordering. Professionals should use these products. Because they contain stainless steel, they also contain nickel. Therefore, Intrace USA assumes no liability for allergic reactions or adverse effects related to material sensitivities. For additional safety data, refer to the FDA guidelines on medical device materials.







Reviews
There are no reviews yet.